June 9th, 2020 (Mississauga, ON) – The CAN Health Network has announced the launch of a promising COVID-19 focused project. Even in these unprecedented times, this project demonstrates that healthcare and the economy have never been more intertwined and showcase the immense ability of Canadian talent.
“Keeping our communities safe by controlling the spread of COVID-19 remains our top priority during these unprecedented times,” said Dr. Dante Morra, Founder of CAN Health Network. “The CAN Health network has enabled companies, like Precision Biomonitoring, to partner with great health care organizations like Sunnybrook Health Sciences Centre and the Research Institute of St. Joe’s Hamilton to develop a rapid COVID-19 test. Important projects like this will help us deal with health challenges like COVID-19 as well as support the regrowth of our economy.”
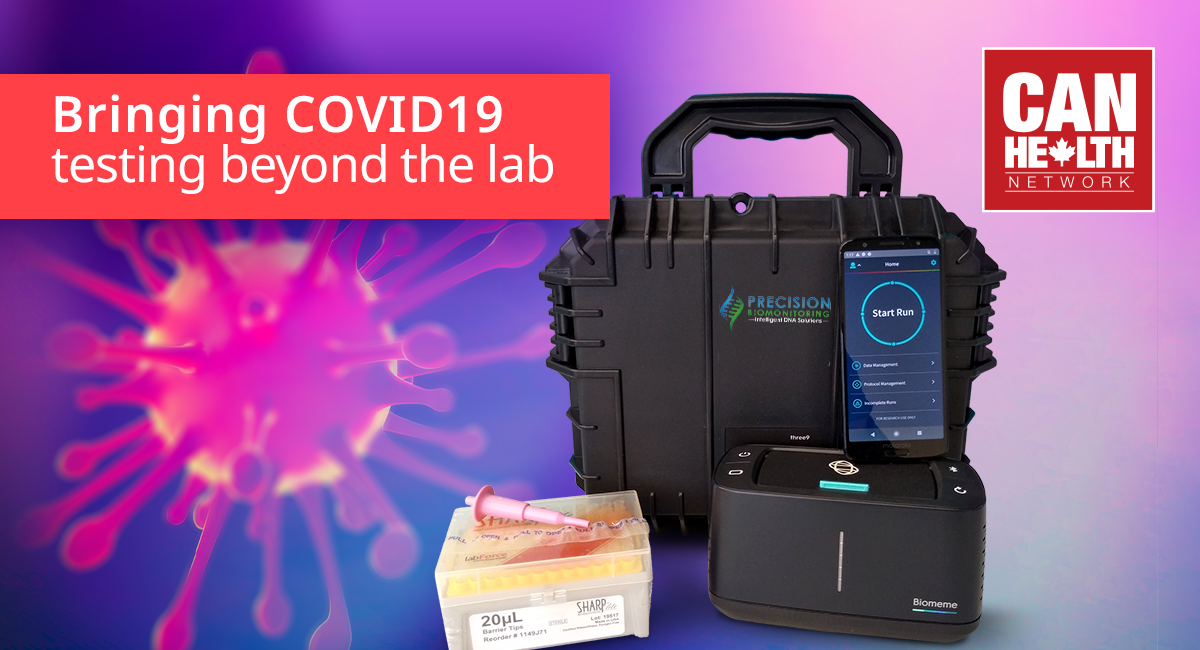
In early April 2020, Precision Biomonitoring Inc. (PBI) partnered with Sunnybrook Health Sciences Centre, a member of the CAN Health Network, to validate a point-of-care COVID-19 diagnostic device. PBI is collaborating with The Research Institute of St. Joe’s Hamilton to implement its existing COVID-19 laboratory-developed test (LDT) for use in PBI’s handheld point-of-care device. The device is ideal for on-the-spot tests, especially when results are needed quickly or where lab testing might be unavailable. Sunnybrook scientists will be helping to validate the new test format alongside St. Joe’s.
“Having a reliable point-of-care test is critical for controlling the spread of COVID-19, especially in community or rural settings where testing may take longer. It will allow these communities to get results faster and will allow healthcare professionals to take the necessary steps and expedite their response to COVID-19 cases,” said Dr. Rob Kozak, clinical microbiologist at Sunnybrook Health Sciences Centre.
PBI, a company founded in 2016 by a team of scientists from the University of Guelph’s Biodiversity Institute of Ontario, has already announced that it will bring its rapid mobile detection device to Indigenous communities in northern Ontario, in response to COVID-19. With the current testing abilities offered, it is taking seven to ten days for a result to be returned to a patient in an Indigenous community.
With PBI’s point-of-care test, results could be obtained by nine patients in as quickly as one hour. This allows for health care providers to make more timely and informed treatment decisions and will enable communities to increase testing speed and capacity.
“Early detection in 60 minutes at the point-of-need is critical to COVID-19 screening efforts to complement central testing, and is absolutely a key as we move forward with efforts to create virus-free zones within remote Indigenous communities and work places in urban areas.” says Dr. Mario Thomas, CEO of Precision Biomonitoring. “The validation of a Canadian-developed test that will be soon manufactured in Canada will contribute immensely to making communities and workplaces safe for vulnerable populations and workers.”
“In the fight against COVID-19, we must harness the talents of both our public and private sectors – and it’s inspiring to see so many Canadians mobilizing,” said the Honourable Mélanie Joly, Minister of Economic Development and Official Languages and Minister responsible for the Federal Economic Development Agency for Southern Ontario. “I want to thank Precision Biomonitoring Inc. for making such important contributions to this endeavour. Improving our testing is a crucial part of our economic reopening and recovery, and this initiative will help us do just that. In this era of uncertainty and upheaval, government, academia and industry are united in our efforts on behalf of all Canadians.”
Quick facts:
- Health-tech represents a $250 billion global market
- Health care spending in Canada is growing at a rate of over 1.5 times our GDP
- PBI mission delivers eDNA surveys that provide the fastest and most accurate results
- CAN Health Network Edges are public or private organizations with shared challenges that form a network to collaborate and adopt innovative solutions.
The CAN Health Network is a Canada-first approach to technology adoption. It will help break down barriers to scaling in the healthcare system and provide an environment for companies to succeed to their full potential. It was created in July 2019, as a result of a federal government investment totaling $7 million, of which the Federal Economic Development Agency for Southern Ontario (FedDev Ontario) and Western Economic Diversification Canada (WD) each contributed $3.5 million. This investment established the Network in Ontario and the West, with expansion to the East, Quebec and the North to come. To learn more about the CAN Health Network, visit www.canhealth.ca.
Media Contact:
For all media inquiries, please contact
Jacqueline Ornelas, Communications & Partnerships Advisor
[email protected] and 416-456-8032
[TheChamp-FB-Comments width=”800″]
Sign up for email
Error: Contact form not found.

